The Drill Down, Issue #11: Basic Primer on Hydrocarbons and Reservoirs
 Introduction
Introduction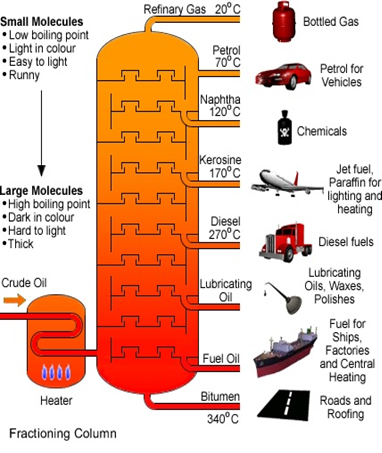
The exploration for oil and gas in any setting is difficult, but the offshore environment especially offers formidable challenges. To have a better understanding of the operations that are undertaken, this Drill Down will be the first of a series providing basic fundamental explanations of the “what, where, and how” of offshore oil and gas exploration, starting with hydrocarbons and hydrocarbon reservoirs.
What is a hydrocarbon?
A hydrocarbon is a chemical “chain” of hydrogen and carbon atoms. These chains vary in length: the longer the chain, the more like a solid, the shorter the chain, the more like a gas. The varieties of hydrocarbons can be utilized for different purposes. For example, a short chain hydrocarbon, such as methane (natural gas), can be used as a fuel for power or household heating. Crude oil contains within it a variety of hydrocarbon chains, spanning from propane (short chain) to gasoline to bitumen/asphalt (long chain). The image to the right depicts the process of separating the various hydrocarbon chains found in crude oil and their uses, shorter to longer chains from top to bottom. (Source: www.minichemistry.com)
 How is it formed?
How is it formed?
In order for a hydrocarbon to form, there must be the right combination of three factors:
-Organic matter (dead plants and animals)
- Time (up to hundreds of millions of years)
- Heat
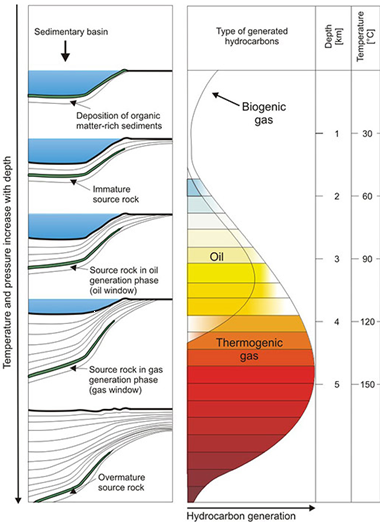
Organic matter is the building block for hydrocarbons. As an example, in an offshore setting, over time a large amount of sea life dies and settles on the seafloor. As rivers pour sediment into the ocean, the sediment begins to bury the organic matter. Over time, significant amounts of sediment are deposited and pressure and temperature increase as the layer is buried deeper and deeper. This causes organic matter to undergo physical and chemical changes, forming hydrocarbons. Temperature and time dictate the “window of opportunity” to form the various hydrocarbon chains. The image to the left portrays how the corresponding temperature with burial depth over time “matures” the organic matter into becoming sources for crude oil and gas generation. (Source: www.infolupki.phi.gov.pl)
Where is it located?
As previously mentioned hydrocarbons are formed by organic matter that was buried and heated deep in the earth for 100s of millions of years. However, as hydrocarbons were formed, they did not become vast, cavernous pools of liquid. As sediment is buried, it undergoes the process of becoming rock, known as lithification, and traps the organic matter in the rock. These rock layers are known as source rock, because they contain the source from which hydrocarbons are formed. The most commonly known source rock is called shale. As the source rock was being formed, the hydrocarbon fluids (liquid and gas) generated by the organic matter were expelled. While some hydrocarbons remained in the source rock, a large amount did not.
So where did the hydrocarbons go?
Although the source rock is where the hydrocarbons were generated, hydrocarbon fluids migrate from the source rock, attempting to move upward due to the significant density difference between hydrocarbons and rocks. But how? As rivers continued to pour and bury layer upon layer of sediment, the sediment eventually lithified and became rock itself. The tiny sediment grains that make up the rock layers do not fit together perfectly, thus microscopic void spaces between the grains exist. These are known as pores. The image to the right is a microscopic image of a rock grain structure. The blue in the image is a resin injected into the rock in a lab to highlight the amount of porosity in a rock. These pores are where hydrocarbon fluids can be found.
Where do hydrocarbons stay?
Different rock types have different porosities, different percentages of pore space within the rock. As hydrocarbons attempt to migrate upward, they will move through porous rock layers and fault lines. Sometimes these fluids reach the seafloor, which are known as natural seeps. However, the fluids may encounter a rock layer that is impermeable. This rock layer is known as a cap rock. The hydrocarbon fluids from the source rock continue to migrate and stop at the cap rock, thus being trapped in porous rock layers below the cap rock. These rock layers are known as reservoir rock would be identified as a hydrocarbon reservoir. Since the most common fluid found in the subsurface is water (brine), it is typical to find a water layer in a hydrocarbon reservoir. This concept is depicted in the bottom right image. (Source www.engineering.com)
Under Pressure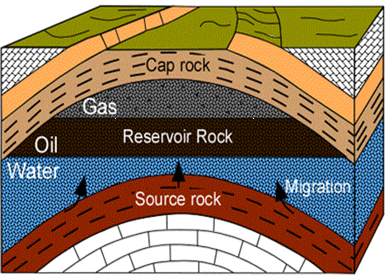
Hydrocarbon reservoirs can be under extreme pressure (tens of thousands of pounds per square inch). All of this pressure (known as pore pressure) is contained in the pores of the reservoir rock. If a hydrocarbon reservoir had a direct conduit to the surface (such as a drilled well), the pore pressure would likely be sufficient to drive reservoir fluids all the way to surface for extended periods of time at very high flowrates (tens of thousands of barrels per day). While drilling a well, uncontrolled hydrocarbon flow to surface is known as a blowout and can have catastrophic consequences to life and property.
Conclusion
This Drill Down provides a foundation for how hydrocarbons are formed and where they can be found. The important takeaways are the right conditions are needed to form hydrocarbons, and hydrocarbons are primarily trapped in porous rock reservoirs thousands of feet below the surface of the earth at extremely high pressures. In the future, we will cover what the oil and gas industry must do in order to explore for hydrocarbon reservoirs.